The spectrophotometer, an instrument used to acquire data using the ELISA. Source: Viv Rolfe via Wikimedia Commons.
One of the first techniques I learned during my Master’s in Biotechnology was how to quantify antibodies or antigens in a sample using the enzyme-linked immunosorbent assay (ELISA).
ELISA remains one of the most powerful and accessible tools used across hospitals, research labs, and public health programs to detect infection.
At its core, it answers a simple question: Has the body encountered a specific pathogen, or is the pathogen itself present in the bloodstream?
During dengue infection, measuring antibody levels helps distinguish between primary and secondary infections.
Likewise during Helicobacter pylori infection, ELISA detects antibodies that indicate exposure, guiding decisions about diagnosis, treatment, and follow up testing.
In this article, we explore why this fundamental technique matters for public health, especially in Latin America, and how ELISA works.
The Origins of ELISA
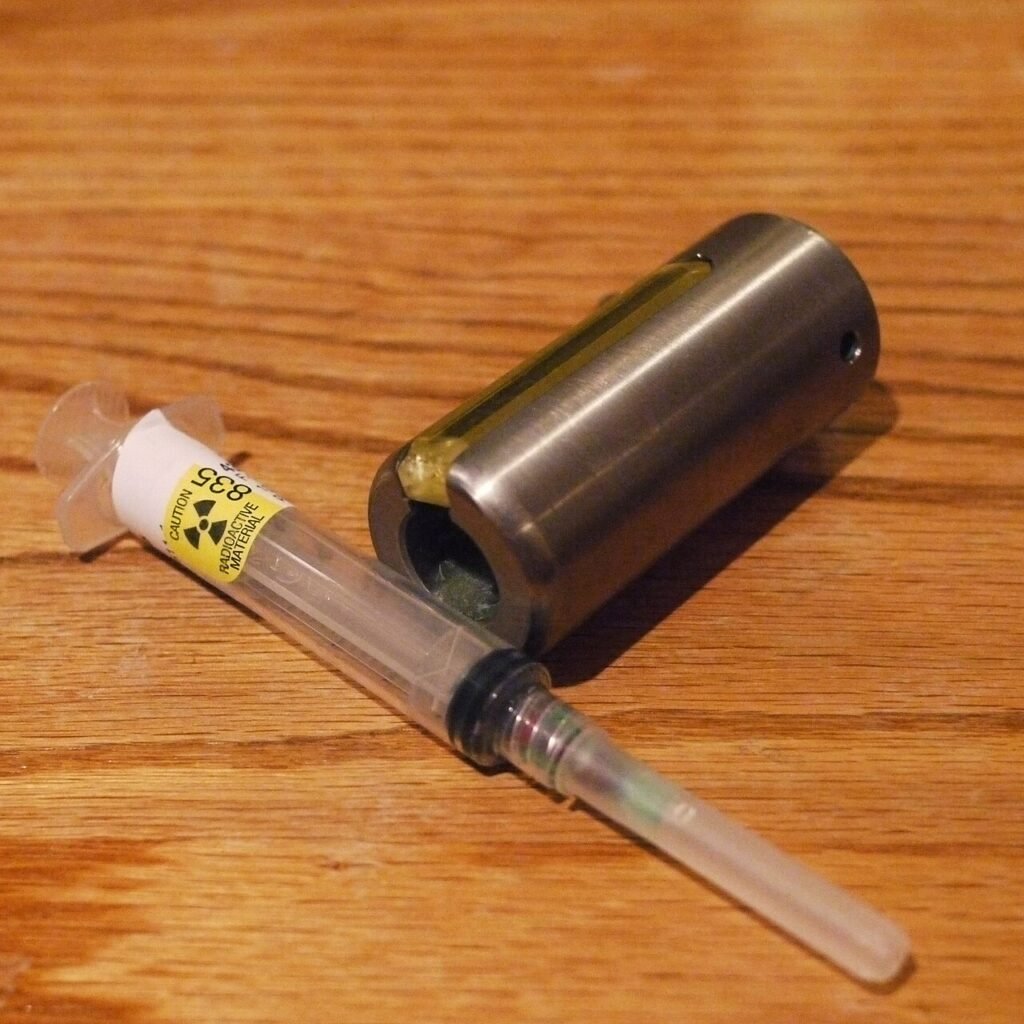
Radioactive syringe. Source: Thirteen of Clubs via Wikimedia Commons.
The ELISA emerged in the early 1970s as a safer, scalable alternative to radioimmunoassays, which relied on powerful but hazardous methods that used radioactive materials.
Researchers Peter Perlmann and Eva Engvall, along with Anton Schuurs and Bauke van Weemen, came in and independently developed methods that replaced radioactive signals with enzyme-linked antibodies to detect antigens or antibodies with high specificity.
By coupling enzymes to antibodies and using colorimetric reactions for readout, they created a technique that was safer, easier to standardize, and adaptable to high-throughput testing.
ELISA rapidly became a cornerstone of diagnostics and epidemiology, enabling large-scale screening for infectious diseases such as HIV and hepatitis.
How ELISA Works
Key Step 1: Coating Wells with Specific Molecules
To understand how the ELISA assay works, it helps to step inside the lab.
The process begins with a plate that contains dozens of tiny wells. Each well acts as a miniature test chamber.
Scientists coat these wells with a capture molecule, often an antigen or antibody, depending on what they are trying to detect.
If the goal is to identify whether a patient has antibodies against a virus, the wells are coated with viral proteins.
If the goal is to detect the virus itself, the wells are coated with antibodies that can bind to that pathogen.
Key Step 2: Adding the Sample and Target Analyte
After blocking the plate to reduce non-specific signal, next comes the patient sample. A small amount of blood serum is added to the wells.
If the target molecule is present, whether antibodies or antigens, it will bind to the coated surface.
This binding step is highly specific. It relies on the same lock and key interactions that drive immune recognition inside the body.
If the target is not present, nothing sticks, and the well remains essentially empty.
Key Step 3: Washing to Remove Background and Unbound Molecules
After giving enough time for binding to occur, the plate is washed.
This step removes any molecules that did not specifically attach.
What remains are only the molecules of interest, anchored to the well through highly selective interactions.
At this stage, the infection signal is present but still invisible.
Key Step Four: Acquiring the Real Signal
To make that signal detectable, scientists add a secondary antibody linked to an enzyme.
This antibody is designed to recognize the target molecule already bound in the well.
If the target is present, the enzyme linked antibody attaches to it, forming a layered complex.
If the target is absent, the enzyme has nothing to bind and is washed away in the next step.
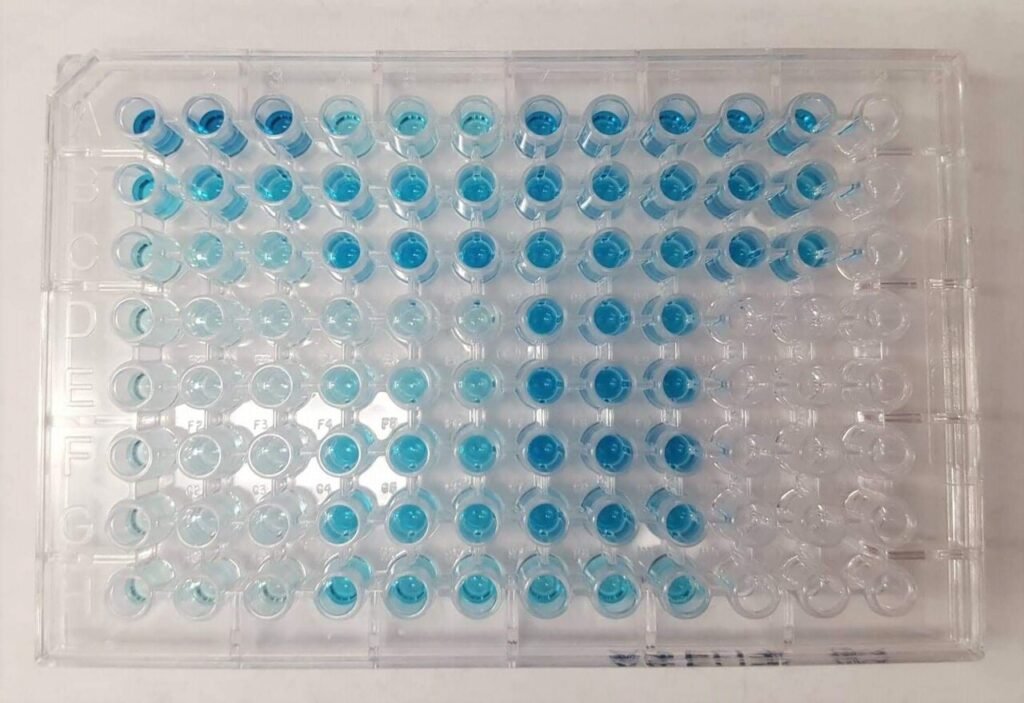
The final step is where ELISA becomes visually powerful.
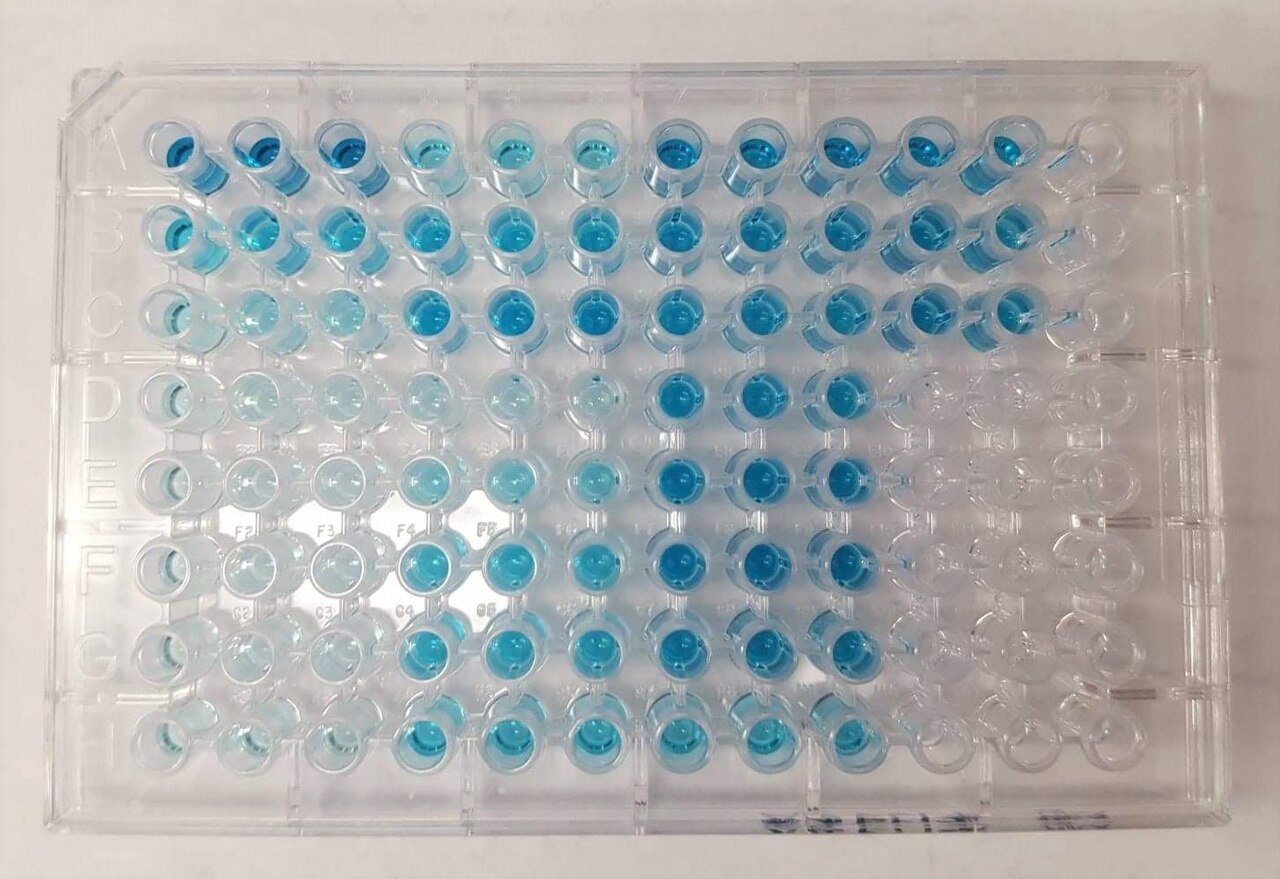
A substrate is added that reacts with the enzyme to produce a color change.
Wells that contain the target molecule develop a visible signal, often shifting from clear to blue or yellow depending on the assay design.
The intensity of this color is not just qualitative. It can be measured using a plate reader.
This allows scientists to quantify how much antibody or antigen is present in the sample.
ELISA Limitations
ELISA assay. Source: Ajpolino via Wikimedia Commons.
ELISA detects antibodies, but the presence of antibodies does not always mean active infection.
This is because antibodies can persist long after the pathogen has been cleared, reflecting past exposure rather than current disease.
Conversely, low antibody levels during early infection leads to false negative results.
Antigen-based ELISAs detect pathogen components directly but often require higher antigen levels to produce a signal.
Overall, ELISA results are most powerful when interpreted alongside clinical symptoms, patient history, and other laboratory findings.
ELISA serves as a first line tool that guides further testing, such as PCR, which can directly detect viral genetic material.
Why ELISA Matters for Public Health

Source: PAHO’s photostream via Flickr.
Inside the lab, ELISA may look like a series of pipetting steps and color changes.
But beyond the bench, it represents a powerful window into how infections move through communities.
Able to scale across large sample sets, it enables health systems to track exposure, identify hotspots, and assess interventions.
Reliable and adaptable biochemical tools like ELISA will continue to remain essential as infectious diseases evolve.
Early detection can change outbreaks, and simple blood tests that read the immune system are central to modern public health.
Subscribe to Pathogenos for more insights that connect lab science to real world disease prevention and public health action.