Source: Whitney Genetics Lab PCR Machine via Flickr.
During my work trying to improve innate immune signaling against viral infection, I often use an attenuated measles vaccine strain as a model virus.
When I infect cells under controlled culture conditions in a dish, an important question immediately arises: how do I know whether the cells are actually infected and whether the virus is replicating?
Viruses, like cells, rely on genetic material (i.e., DNA, RNA) to function. But unlike cells, viruses can only express those genes after they infect a host cell and hijack its machinery to produce viral components.
When we zoom out from the lab to the human body, the same principle applies. Respiratory viruses such as influenza infect the epithelial cells lining our airways and begin expressing their genes as they replicate.
Scientists use this biological signal to measure infection. If a virus is replicating, its genes must be present and expressed. The challenge therefore becomes detecting and quantifying those viral genes accurately.
The quantitative polymerase chain reaction (qPCR) solves this problem by detecting extremely small amounts of viral genetic material with remarkable precision.
For decades, PCR-based diagnostics have anchored infectious-disease detection worldwide because they can identify viral material quickly and reliably, often within hours.
Understanding how qPCR works reveals something bigger than a laboratory technique. It shows how molecular biology translates directly into tools for detecting infection and protecting public health.
What is qPCR?
When doctors confirm a viral infection, the answer often comes from qPCR.
At its core, qPCR works by amplifying genetic material. Viral genomes present in extremely small amounts inside a patient sample are copied repeatedly until they become measurable.
What begins as only a few molecules of viral RNA can be transformed into billions of detectable copies. This amplification process allows scientists to detect viruses even when only trace amounts are present in a sample.
Many viruses that infect humans, including SARS-CoV-2, carry RNA genomes rather than DNA. Because PCR can only copy DNA, scientists first convert viral RNA into DNA through a step known as reverse transcription.
Once this conversion occurs, short DNA fragments called primers bind to specific regions of the viral sequence. These primers guide enzymes that build new DNA strands while fluorescent markers attach to the growing molecules.
The result is a diagnostic system capable of detecting infection directly at the molecular level.
How Laboratories Detect Virus in Blood
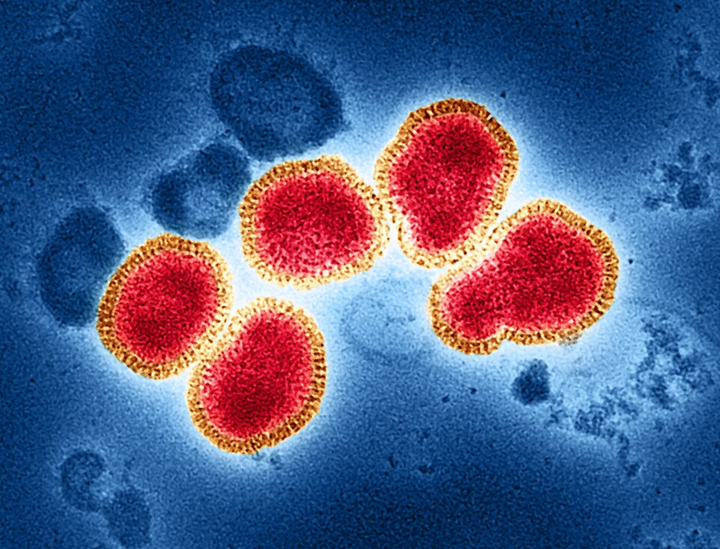
Influenza A virus. Source: NIAID via Wikimedia Commons.
The process begins with sample preparation, where clinical samples are treated with chemical solutions that break down cellular components and isolate RNA.
f infection is present, this RNA mixture contains both human genetic material and viral RNA, making extraction the first critical step in pathogen detection.
Next comes reverse transcription, where viral RNA is converted into DNA. Because PCR amplifies DNA rather than RNA, an enzyme called reverse transcriptase generates complementary DNA that can serve as the template for amplification.
The reaction then enters the PCR amplification stage inside a thermal cycler. The instrument repeatedly heats and cools the reaction, allowing DNA to replicate.
Each cycle doubles the genetic material (two copies become four, four become eight) leading to billions of DNA copies after roughly 35 cycles.
As amplification proceeds, fluorescent markers bind to newly synthesized DNA. Instruments measure fluorescence after each cycle, and when the signal crosses a defined threshold, viral genetic material is detected.
The number of cycles required to reach this threshold becomes one of the most informative measurements in the entire assay.
Understanding Viral Load: The Ct Value
One of the most important outputs of qPCR is the cycle threshold, or Ct value. The Ct represents the number of amplification cycles required before viral genetic material becomes detectable above background fluorescence.
If large amounts of viral RNA are present at the start of the reaction, the fluorescent signal appears quickly and the Ct value is low.
If only small amounts of viral material are present, more amplification cycles are required before detection occurs, resulting in a higher Ct value.
In simple terms, the faster the signal appears, the more virus was likely present in the original sample.
Because Ct values reflect the starting quantity of viral genetic material, researchers often use them as a proxy for viral load, which refers to how much virus is present in the body.
Viral load measurements can provide insights into infection dynamics and disease severity.
However, Ct values must always be interpreted carefully. Differences in sample collection, laboratory calibration, and the stage of infection can influence the results.
For this reason, clinicians combine molecular data with clinical observations when interpreting diagnostic tests.
Why qPCR Became the Diagnostic Gold Standard

Source: pexel
Reverse-transcription qPCR has become the gold standard for detecting many viral infections, including COVID-19. The technology offers a combination of sensitivity, speed, and reliability that few other diagnostic methods can match.
The technique can detect extremely small amounts of viral genetic material, allowing infections to be identified even at early stages.
Results can often be delivered within a few hours, enabling rapid clinical decision-making. In addition, modern real-time PCR systems use closed reaction tubes that reduce the risk of contamination during analysis.
Compared with traditional virus isolation methods, which may require days of cell culture, qPCR provides a faster and more scalable approach to pathogen detection.
Over the past two decades, the technology has supported the diagnosis of major infectious threats, including Ebola, Zika, MERS, and SARS.
An Important Limitation Scientists Consider
Despite its strengths, qPCR has an important limitation that scientists must consider when interpreting results.
The technique detects active infection, meaning it identifies viral genetic material that is currently present in the body. Once the virus has been cleared, the genetic material is no longer detectable.
To determine whether someone was infected in the past, clinicians often rely on antibody testing, which measures the immune system’s response to a virus rather than the virus itself.
Combining molecular diagnostics with immune-based tests provides a more complete picture of disease exposure and spread.
Understanding these differences is essential for responsible diagnostic interpretation and effective public-health surveillance.
Why Measuring Viral Load Matters for Public Health
Doctor looks at boy’s throat in medical examination. Mexico. Source: Curt Carnemark / World Bank via Flickr.
Quantifying viral load does more than confirm infection. It also provides information that can guide medical and public-health decisions.
Viral load measurements help clinicians assess disease progression and evaluate whether treatments are working.
Epidemiologists use these measurements to better understand how viruses spread through populations and to identify individuals who may be more likely to transmit infection.
By enabling faster detection and more precise measurement of viral activity, molecular diagnostics strengthen outbreak response systems and support more effective containment strategies.