
Source: EmanuelArgentino1982 via Wikimedia Commons.
Highlights
- Latin America’s climate drives dengue via Aedes mosquitoes
- Two dengue vaccines exist and Qdenga offers broader use
- Vaccine access uneven and many rely on private markets
- Dengue control needs vaccines vector control and surveillance
Latin America’s climate makes it a hotspot not only for tourists like you and I looking to connect with the sun, but also for something far less welcome: Aedes mosquitoes carrying arboviruses like dengue, Zika, and chikungunya.
With millions of dengue cases reported each year, the region has become a critical testing ground for vaccination strategies.
The plot twist is that, despite decades of research, only two dengue vaccines remain available globally, and their distribution across Latin America varies widely.
Understanding which vaccines are available, and where they are actually being deployed, is essential for translating science into real public health action.
The Two Dengue Vaccines: Dengvaxia and Qdenga
Source: Renato Alves/Agência Brasília via Wikimedia Commons.
Today, two licensed dengue vaccines exist: Dengvaxia (CYD-TDV) and Qdenga (TAK-003).
Both are tetravalent, live-attenuated vaccines.
“Tetravalent” means these vaccines are designed to protect against all four dengue virus serotypes (DENV-1 to DENV-4). “Live-attenuated” means they use weakened (mutated) versions of the virus to trigger an immune response without causing full disease.
Dengvaxia
Dengvaxia was the first dengue vaccine introduced globally in 2015 developed by Sanofi.
The catch is that its use is highly restricted, where it is only recommended for people with confirmed prior dengue infection like children living in endemic areas.
This is because clinical studies have shown that without prior dengue infection, dengvaxia vaccination could increase the risk of severe disease due to what is known as antibody-dependent enhancement (ADE).
This side-effect has significantly limited its large-scale public health use.
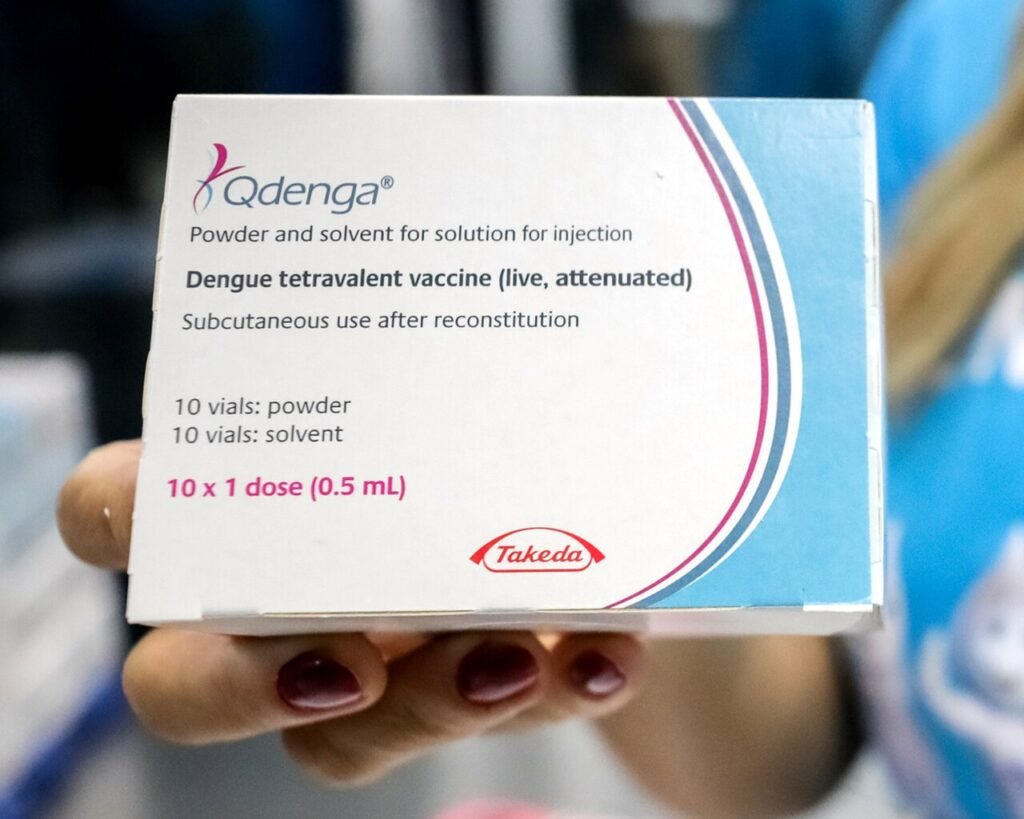
Qdenga
In contrast, Qdenga (TAK-003) represents a second-generation approach.
Developed by Takeda, it can be used regardless of prior infection status, making it far more practical for mass vaccination campaigns.
The vaccine is administered in a two-dose schedule spaced three months apart and is recommended by the World Health Organization (WHO) for children aged 6–16 years in high-transmission settings.
This difference, restricted vs. broadly applicable use, is shaping how dengue vaccination is unfolding across Latin America.
Where Dengue Vaccines Are Used in Latin America
Source: Governo do Estado de São Paulo via Wikimedia Commons
Brazil: A Regional Leader in Deployment
Brazil has emerged as a key player in dengue vaccination.
Both Dengvaxia and Qdenga have been licensed, but Qdenga is becoming the preferred option due to its broader safety profile.
Qdenga was approved in Brazil in 2023 and quickly entered distribution, including public health discussions around national immunization strategies.
Brazil is also a hub for vaccine research and future development, with additional dengue vaccine candidates undergoing late-stage trials in the country.
Argentina and Colombia: Expanding Access

Source: Tom Fournier via Pexels.
Argentina and Colombia have both approved Qdenga, with availability beginning in 2023–2024, primarily through private markets and expanding toward broader use.
In Colombia, where dengue is endemic and outbreaks are frequent, vaccination is being considered as part of integrated vector-control strategies.
The country’s approval reflects growing regional confidence in second-generation vaccines.
Mexico, Peru, and Central America: Mixed Approaches
Countries such as Mexico, Peru, Costa Rica, and Guatemala have historically relied on Dengvaxia, which was introduced between 2015 and 2016.
However, Dengvaxia’s limitations, particularly the need for pre-vaccination screening, have slowed widespread adoption.
As a result, many of these countries are now transitioning toward evaluating or introducing Qdenga as a more scalable solution.
Regional Trends: Private vs Public Access
A key challenge across Latin America is how vaccines are distributed.
In many countries, Qdenga is currently available primarily through private healthcare markets, meaning access depends on ability to pay.
At the same time, WHO prequalification of Qdenga has opened the door for procurement through international organizations such as PAHO and UNICEF, which could expand access in lower-resource settings.
Why Distribution Remains Uneven

Source: Luis Perez via Wikimedia Commons.
Despite scientific progress, dengue vaccine coverage across Latin America remains uneven. Several key barriers explain why:
1. Safety-driven policy restrictions
Dengvaxia’s requirement for prior infection testing adds logistical complexity, limiting rollout in many countries.
2. Cost and infrastructure
Vaccines like Qdenga require cold-chain distribution and multiple doses, which can strain health systems already managing dengue outbreaks.
3. Public trust and vaccine hesitancy
Past controversies surrounding dengue vaccination campaigns have influenced public perception, especially in regions where trust in health institutions is fragile.
4. Integration with vector control
Vaccination alone is not enough. Countries must integrate vaccines with mosquito control strategies, surveillance, and public education.
Why This Matters for Public Health in Latin America

Source: World Bank Photo via Flickr.
The emergence of Qdenga marks a turning point. For the first time, a dengue vaccine can be deployed without requiring prior infection screening, making population-level prevention more realistic.
However, vaccination is not a silver bullet.
Dengue transmission is driven by climate, urbanization, and vector ecology, all of which are intensifying across Latin America.
This means the future of dengue control will depend on layered interventions vaccination, vector surveillance and control, climate-informed risk prediction, and community level prevention strategies.
The Takeaway
Latin America is at the forefront of dengue vaccine implementation. While Dengvaxia laid the groundwork, its limitations restricted its impact.
Qdenga is now reshaping the landscape, with growing adoption in countries like Brazil, Argentina, and Colombia.
But access remains uneven, and large-scale impact will require coordinated policy, funding, and public health infrastructure.
Dengue vaccines are no longer theoretical, they are here. The challenge now is ensuring they reach the populations that need them most.




